RESEARCH ARTICLE
Polysorbate 80 Coated Solid Lipid Nanoparticles for the Delivery of Temozolomide Into the Brain
Pawan Yadav1, Goutam Rath1, Gazal Sharma1, Ranjit Singh2, Amit Kumar Goyal1, *
Article Information
Identifiers and Pagination:
Year: 2018Volume: 8
First Page: 21
Last Page: 28
Publisher Id: TOPHARMJ-8-21
DOI: 10.2174/1874143601808010021
Article History:
Received Date: 22/02/2018Revision Received Date: 03/10/2018
Acceptance Date: 14/10/2018
Electronic publication date: 30/10/2018
Collection year: 2018
open-access license: This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International Public License (CC-BY 4.0), a copy of which is available at: (https://creativecommons.org/licenses/by/4.0/legalcode). This license permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction:
Anti-angiogenic therapy can produce transient regression in tumor in case of Glioblastoma (GBM); however, no prolongation of patient survival rate had so far been achieved.
Methodology:
To address this problem, an effort was made to design and characterize a temozolomide loaded nanosystem for targeting the tumor vasculature in the brain using polymeric nanoparticles. It included the formation of Temozolomide (TMZ) loaded Solid-Lipid Nanoparticles (SLNs) and their conjugation with polysorbate-80 (P-80) which enhanced the penetration of drug to blood-brain barrier resulting in the enhancement of pro-apoptotic activity.
Results:
Conjugating nanoparticles with a tumor-penetrating polymer (P-80) further enhanced the therapeutic efficacy of the drug.
Conclusion:
The animal studies indicated the enhanced potential of the developed system in the effective treatment of glioblastoma.
1. INTRODUCTION
Brain cancer is one of the prominent dreadful diseases among other CNS related disorders. Although various treatment strategies including surgery, chemotherapy have been available to control the progression of diseases, but still mortality rate against brain cancer is very high [1, 2]. This may be attributed to the development of resistance against existing treatment strategies [3]. There is an unmet need to provide better strategies especially to blood-brain barriers that provide enough quantity of drugs to the site of action. It is very difficult to improve the availability of drugs to the brain compartment by passing blood-brain barriers [4-8].
Many novel drug delivery systems have been designed to improve the bioavailability of drug to brains including nanoparticles, micellar systems, microemulsion, solid lipid nanoparticles, liposomes, niosomes etc. [9-12]. Among them, nanoparticles based delivery of drugs to the brain has shown significant potential. Nanometric size of these particulate carriers not only improves the permeation and availability of drugs to the brain, but also provides a large surface area, improves solubility and ability to encapsulate both hydrophilic and lipophilic drugs. Further, it is also possible to regulate the release of drugs for a prolonged period of time [13, 14]. Furthermore, several studies have reported that the presence of surface active agents, permeabilizers, solubilizers cationic and anionic agents at the surface of nanoparticles provides better stability of carrier systems in blood and better brain uptake. Many drugs have been delivered using these strategies that include paclitaxel, doxorubicin, docetaxel, dalargin, methotrexate, kytorphin, loperamide, tubocurarine, MRZ2/576 etc. [15-23].
Temozolomide, a prodrug, is a nonfunctional alkylating agent that readily crosses the blood-brain barrier. It is derived from a series of modified imidazotetrazinones. It is chemically related to dacarbazine and is the 3-methyl derivative of the experimental anticancer drug mitozolomide [24, 25]. Unlike dacarbazine, temozolomide does not require hepatic metabolism to the intermediate species methyltriazen-1-yl imidazole- 4-carboxamide (MTIC). Chemical stability studies have shown that temozolomide spontaneously hydrolyzes to MTIC above pH 7 [22]. Many studies have demonstrated the chemotherapeutic potential of this drug in various tumor models [26-28]. Further, temozolomide not only demonstrated broad-spectrum anti-cancer activity, but also provided better pharmacokinetic profile including better penetration in the brain with relative low toxicity compared with its parent compound mitozolomide [29-37]. In the present studies, surfactant coated lipid nanoparticles were explored for the delivery of temozolomide. Further, its pharmacokinetics was also compared with an uncoated lipidic nanoparticulate formulation for providing better availability of drugs to the brain compartments.
2. MATERIALS AND METHODS
2.1. Materials and Instruments
Temozolomide was obtained as a gift sample from Cipla Pharmaceuticals Banglore. Tripalmitin, soya lecithin and polysorbate 80 (Tween 80) were purchased from Sigma Pvt. Ltd. New Delhi. All other materials and reagents used in the study were of analytical/HPLC grade. Several instruments have been used during the development of solid lipid nanoparticles including double beam UV spectrophotometer, digital balance, incubator cum shaker, particle size and zeta-potential analyzer, scanning electron microscope, transmission electron microscope, HPLC etc.
2.2. Development of Solid Lipid Nanoparticles
Solid lipid nanoparticles were prepared by partially modified hot homogenization method [31]. Initially, temozolomide (3mg) and tripalmitin (30mg) were dissolved in a mixture of ethyl acetate and methanol (1:1), subsequently resultant solution was placed under rotatory evaporator for complete removal of organic solvent under inert atmospheric condition. Resultant dried drug-lipid mixture was further melted at 50oC ± 5oC for 15 min. Thereafter, 40 ml of a hot aqueous solution of Poloxamer 188 was further homogenized with molten lipid phase at 4000 rpm at 50oC ± 5oC temperature condition. Emulsified homogenized lipid formulation was further subjected under sonication using a probe sonicator at 50W for 20 min. The obtained nanoemulsions were cooled at 6oC to affect re-crystallization for the formation of solid lipid nanoparticles.
2.3. Coating of Surfactant on Solid Lipid Nanoparticles
Surfactant coated solid lipid nanoparticles were prepared by using polysorbate 80 (Tween® 80). 1% (w/v) solution of polysorbate 80 was admix nanoparticle suspension for surface coating of nanoparticles under magnetic stirring for 30 min. Resultant coated nanoparticles were centrifuged, re-dispersed and were stored under refrigerated condition for further characterization [32].
2.4. Size, Shape and Zeta Potential Determination
Size and zeta potential of developed nanoparticles were determined by zetasizer (DeslananoTMC, instrument, Beckman Coulter, USA). Similarly, SEM and TEM techniques were also used to determine the size and shape of developed nanoparticles.
2.5. Entrapment Efficiency
Entrapment efficiency of developed different SLN formulations was analyzed as reported by Venkateswarlu and Manjunath, with minor modifications [33]. After the development of solid lipid nanoparticles, resultant dispersion was taken in centrifuge tube and centrifuged until the separation of solid lipid nanoparticles from dispersion. Finally, the amount of unentrapped drug was measured in the supernatant. The percent EE was calculated using the following formula.
% Entrapment Efficiency =

2.6. In Vitro Drug Release Studies
In vitro drug release kinetics of developed formulation was performed by diffusion method. Drug-loaded SLNs and TMZ-SLNs suspensions were filled in dialysis tubes (12,000 molecular weight cutoff) and tagged tightly with the tube and subsequently, formulation filled dialysis tube was placed into a beaker containing 200 ml phosphate-buffered saline (pH 6.8) under a magnetic stirrer at 37oC. During the in-vitro release studies, 0.5 ml of the sample was collected at a predefined time interval and the same amount of fresh medium was added. All the collected samples were further diluted appropriately and analyzed by UV spectrometry.
2.7. Cell Viability Assay
The toxicity of developed formulation was determined by MTT assay as reported previously [34]. 96 wells microtiters plates were used to grow cells at 2x104 cells/ 200µl growth medium /well. Finally, cells were incubated with fresh growth medium containing different formulations in the concentration range of 2 -14µg/ml. Later, growth medium was aspirated and washed with 200µl PBS (1M, pH 6.8). Afterward, MTT reagent (0.8 mg/ml) was added in each well and incubated for 5 h. Later, the supernatants were discarded and DMSO was added in each well and again incubated at 37ºC for 1 hour, and finally, absorbance was recorded using ELISA reader. An OD value of control cells (unexposed cells) was taken as 100% viability.
2.8. Pharmacokinetics and Tissue Distribution
Six healthy mice (provided by IIM Jammu, body weight 20±1.0/kg, three females and three males) were taken for biological studies. The mice fasted overnight with free access to water. The different formulations were injected intravenously (10 mg/kg body weight) and blood samples from different groups were added in heparinized tubes at predefined time intervals of 0, 0.25, 0.5, 1, 2, 4, 6, 8, 12, 24, 36 and 48 hr and centrifuged at 2000 rpm for 15 min. Further, plasma were collected after centrifugation at 2000 rpm for 15 min and stored at -20oC until use. Availability of drug within brain has also been determined after scarifying mice. Brain and its tissue were dissected and further processed as discussed previously. Collected plasma and biological samples were further processed and used for determination of drug content at a different time interval via HPLC analysis.
3. RESULTS
Temozolomide is the newly approved drug by the FDA for the treatment of malignant glioma. The present study explores the targeting potential of an anticancer drug (temozolomide) via delivery through lipidic nanoparticles. The developed system would be able to avoid the RES system of the mononuclear in the brain using ear phagocytes systems. Temozolomide loaded solid lipid nanoparticles were prepared by hot melt homogenization method in conjunction with sonication.
Solubility of the drug was determined in various solvents at room temperature. Temozolomide was found to be freely soluble in methanol, chloroform, slightly soluble in ethanol, acetone and insoluble in water. These observations showed that the drug is highly lipophilic in nature. Melting range of the drug was found to be 210-213oC, which is in agreement with the reported values. The methanolic solution of temozolomide exhibited absorbance maxima at 328nm which is concordant with I.P. standards. The IR spectrum of drug samples was found to be in concordance with the reference spectrum given in IP 2010 as the observed peaks were very close to standard peaks. All these observations confirmed the identity of temozolomide. The standard curves of temozolomide (triplicate) were prepared in methanol and PBS (pH 6.8). The absorbance data obtained was subjected to linear regression. The correlation coefficient was found to be 0.998, indicating good linearity.
During the preparation of solid lipid nanoparticles, formulations were optimized by using different lipid ratio subjected to drug entrapment efficiency and particle size analysis. Increasing the methanol: ethyl acetate ratio, the size of SLNs’ was noted to be decreased viz. 163.3±3.2nm (D1), 162.3±3.6nm (D2), 146.7±3.9nm (D3), 151.8±4.3nm (D4). It was noted that on increasing the amount of soya lecithin, entrapment efficiency decreased, 71.46±1.4% (C1), 70.56±1.3% (C2), 70.93±1.5% (C3), 69.80±1.3% (C4). The potential drop was also observed on increasing homogenization time, 28.4±2.3 (A1), -34.7±2.3 (A2), -33.08±3.1 (A3), -40.09±2.1 (A4). D3 formulation was found to be optimum out of sixteen developed formulations, having size 146.7±3.9nm, PDI 0.314±0.003, zeta potential -38.27±2.4mV, mobility -2.984±0.01e-004cm2/Vs, entrapment efficiency 72.90±1.3% Table 1.
| S.NO | Parameter | Value |
|---|---|---|
| 1 | Size | 146.7±3.9nm |
| 2 | PDI | 0.314±0.003 |
| 3 | Zeta potential | -38.27±2.4mV |
| 4 | Mobility | -2.984±0.01e-004cm2/Vs |
| 5 | Entrapment efficiency | 72.90±1.3% |
The shape of the SLNs was examined using SEM & TEM. SEM & TEM studies of all the solid lipid nanoparticles were performed. The images revealed that the SLNs were spherical in shape and homogeneously distributed (Figs. 1 and 2).
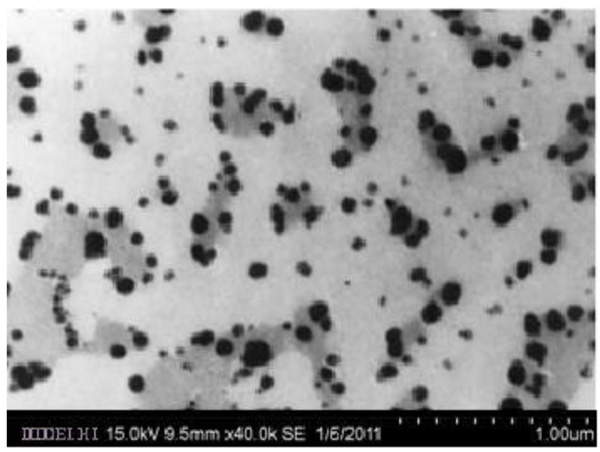 |
Fig. (1). Scanning electron microscopy of solid lipid nanoparticles. |
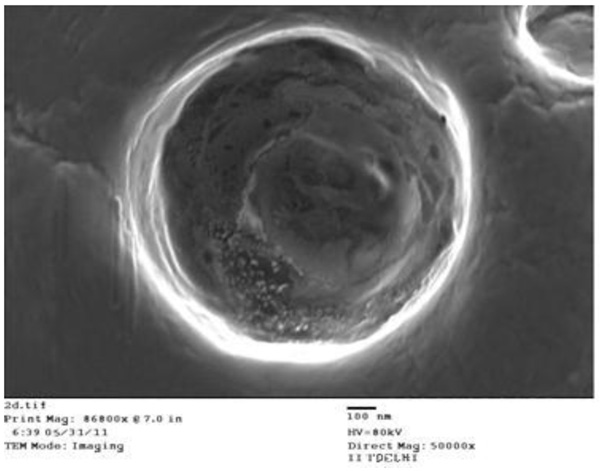 |
Fig. (2). Transmission electron microscopy of solid lipid nanoparticles. |
In vitro drug release of different formulations was evaluated by diffusion method in phosphate-buffered saline (pH 6.8) at 37°C until the complete release of drugs from the formulations. P-80 coated SLNs exhibited 82.46 ± 1.43% drug release and followed first order kinetics. The P-80 coated TMZ SLNs showed slower drug release as compared to TMZ SLNs. The drug release studies are shown in (Fig. 3).
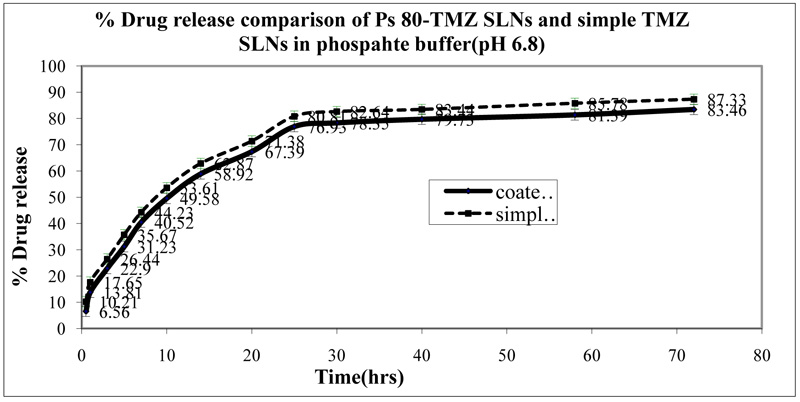 |
Fig. (3). In-vitro drug release studies of the P-80 coated temozolomide loaded solid lipid nanoparticles and plain temozolomide loaded solid lipid nanoparticles. |
The comparative cytotoxicity studies of different coated and uncoated SLNs’ formulations were carried out using MTT assay. Percentage cell viability of formulation in U373MG glioblastoma cancer cell lines was evaluated. Fig. (4) presents the IC50 value for different formulations. The IC50 value was found to be 8µg/ml with polysorbate 80 (P-80) coated solid lipid nanoparticles. The percent (%) drug release from the P-80 coated solid lipid nanoparticles was a bit lesser than the uncoated SLN which may be attributed to the P-80 coating over the solid lipid nanoparticles.
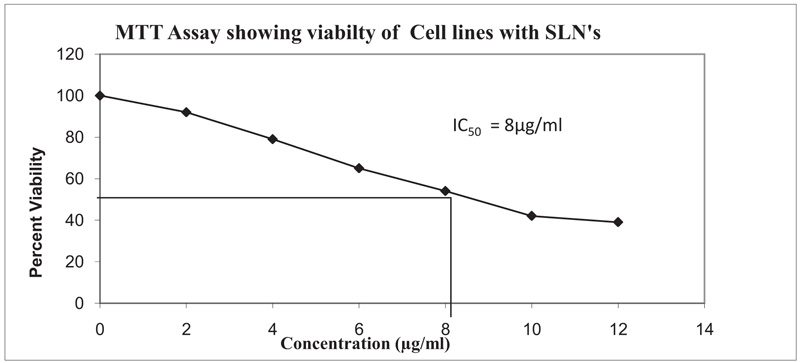 |
Fig. (4). Percent cell viability of P-80 coated Temozolomide loaded solid lipid nanoparticles in U373MG glioblastoma cancer cell lines using MTT assay. |
Various pharmacokinetic parameters such as MRT (h), AUC (h ng ml-1), C max (ng ml−1), CL (mg kg−1/h ng ml−1) were calculated and compared with the free drug Table 2. In plasma, the AUC and MRT were increased for P-80 coated TMZ SLNs’ as compared to other formulations. The concentration of temozolomide in the brain was significantly increased in case of P-80-TMZ SLNs as compared to TMZ solution & TMZ SLNs’ (26.19±0.11, 6.43±0.21 & 15.07±0.21% respectively). The results showed that polysorbate 80 coated SLNs exhibited better delivery of the drug in the brain compared to other formulations.
| S.No. | Parameter | TMZ Solution | TMZ SLNs’ | P-80 coated SLNs’ |
|---|---|---|---|---|
| 1 | MRT (h) | 3.78±0.03 | 45.32±0.82 | 57.89±0.45 |
| 2 | AUC (h ng ml-1) | 807.63±2.4 | 7305.67±4.2 | 9605.84±3.7 |
| 3 | C max (ng ml−1) | 376.2±3.5 | 200.1±3.9 | 157.66±2.4 |
| 4 | CL (mg kg−1/h ng ml−1) | 0.0124±0.0001 | 0.0014±0.0001 | 0.0006±0.0001 |
Several studies have also supported that surfactant coated nanoparticles are an important tool to increase the uptake of drugs into the brain. This may be attributed to the interaction of surface active agents with the endothelial microvasculature of the brain. In addition to this, the presence of surfactant may help loose the tight junction of the BBB. This may lead to better uptake of surfactant coated nanoparticulate formulation.
4. DISCUSSION
The blood-brain barrier is the highly selective tight junction defence system to prevent the passage of unwanted harmful substances [13]. This phenomenon of BBB is a major limiting factor of the absorption of drugs within the brain tissue. Several approaches have been developed to increase the permeability of drugs across the brain especially nanotechnology [37]. Furthermore, the presence of surfactant over nanoparticles has also demonstrated increased uptake of the drugs. Polysorbate 80 coated solid lipid nanoparticles have shown significantly better uptake of drugs compared with other formulation. Pharmacokinetic parameters like increased values of AUC and MRT have highlighted the important of surfactant coating over nanoparticle surface. The concentration of drug in the brain was significantly higher in case of polysorbate-80 coated nanoparticles as compared to TMZ solution & simple TMZ SLNs. The results proved the potential of coated nanoparticles in enhancing the delivery of a drug across the brain.
CONCLUSION
The polysorbate-80 coated SLNs are multipurpose nanoparticulate systems able to deliver both hydrophilic and lipophilic drugs and also avoiding RES uptake to lesser uptake of the drug to other organs. The developed formulations may also reduce the total dose required for the therapy with a concurrent reduction in dose-related toxicity. However, further studies are required to endorse their potential as an effective drug delivery system.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
All experimental procedures and protocols used in this investigation were reviewed and approved by the Institutional Animal Ethics Committee (IAEC), I.S.F. College of Pharmacy, Moga, Punjab, India.
HUMAN AND ANIMAL RIGHTS
No humans were used in the experiments. All the reported experiments involving animals used in the study were in accordance with the guidelines of the council for the purpose of control and supervision of experiments on animals (CPCSEA).
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The authors are thankful to Chairman, ISF College of Pharmacy, Moga Punjab, India, for providing necessary facilities to carry out this work.




